Home
COVID-19 Testing for Healthcare Providers


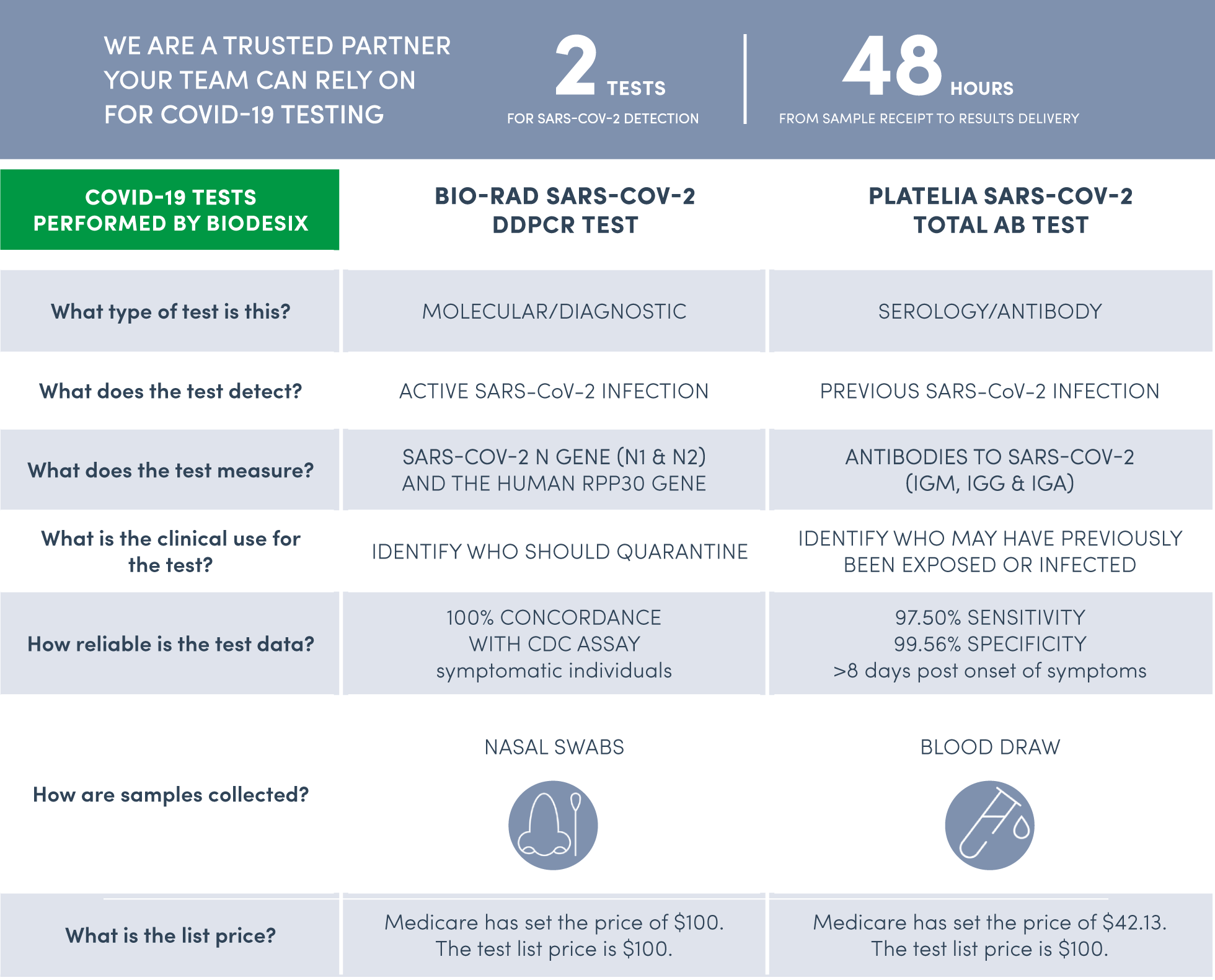
In response to the global pandemic, we have commercialized the Biodesix WorkSafe™ COVID-19 Testing Program, which includes two EUA-FDA authorized tests (molecular & serology) for the SARS-CoV-2 virus in partnership with Bio-Rad Laboratories.
We offer an end-to-end customized testing solution inclusive of specimen collection kits, coordinated logistics, and billing support.
We operate two high-complexity CAP/COLA accredited, CLIA and NYS CLEP certified laboratories with high capacity for COVID-19 testing and results reported within 48 hours on average.
We recognize everyone’s COVID-19 situation is unique, which is why we wanted to provide you with a list of customizable steps in our COVID-19 testing program to fit your needs.
COVID-19 Tests – option to order both the molecular and antibody test together or individually depending on your testing needs.
Specimen Collection Kits – we provide nasal swabs and blood collection kits for molecular and antibody testing, respectively.
Mobile Specimen Collection – we can facilitate scheduling a healthcare provider to perform centralized specimen collection (e.g., testing at your facility or at home as needed).
Scheduled Courier Pick-Up (Colorado & Kansas) – we can ensure same-day specimen pick-up with scheduled local couriers.
FedEx Overnight Shipping (US) – we use FedEx for overnight shipment of specimens to the Biodesix laboratory.
Open All Week – we accept specimens for testing 7-days per week to ensure we deliver results as soon as possible.
Individual & Batch Test Reporting –we offer two options for test result reporting that are HIPAA compliant.
COVID-19 Demographic Report – we can provide a demographic coversheet for a snapshot of the impact to your organization.
Biodesix is able to bill you, except as required by State or Federal regulations, or the patient’s insurance for COVID-19 testing. The list price for each test is $100.
A leading data-driven diagnostic solutions company helping answer critical clinical questions faced by physicians, researchers, and biopharmaceutical companies, with a primary focus in lung disease.
CORPORATE HEADQUARTERS
919 W. Dillon Rd. | Louisville,
CO 80027
Phone (303) 417-0500
Fax (866) 432-3338
© Copyright 2024 Biodesix.
All Rights Reserved.